
Support Growth and Reduce Risk
with Electronics Design

Innovate Faster
with Collaboration
- Grow cross-domain collaboration & reduce design cycle-time
- Accelerate innovation by leveraging design reuse & best practices
- Build agility with information sharing between engineers & supply chain

Support Compliance
with Excellence
- Create transparency & traceability that extends from designers to sustainment
- Innovate new products with verification, validation and change management for compliance
- Manage centralized component selection with approved suppliers

Keep Pace with
System-Level Control
- Increase control while simplifying workspace access & data management
- Protect with enterprise security and role-based access controls
- Standardize intelligent systems & processes to accelerate product launches



Engineer for Complexity and Precision
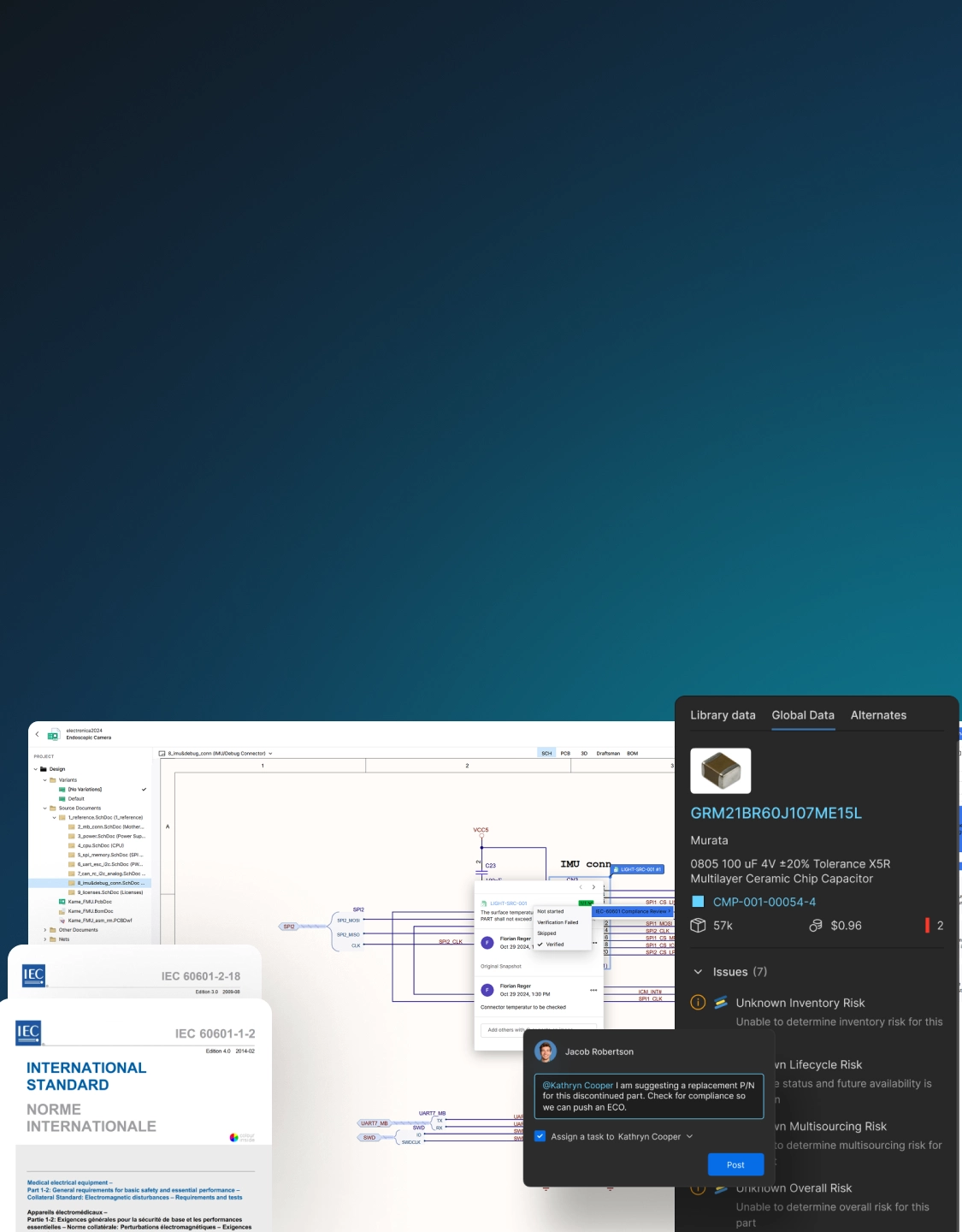
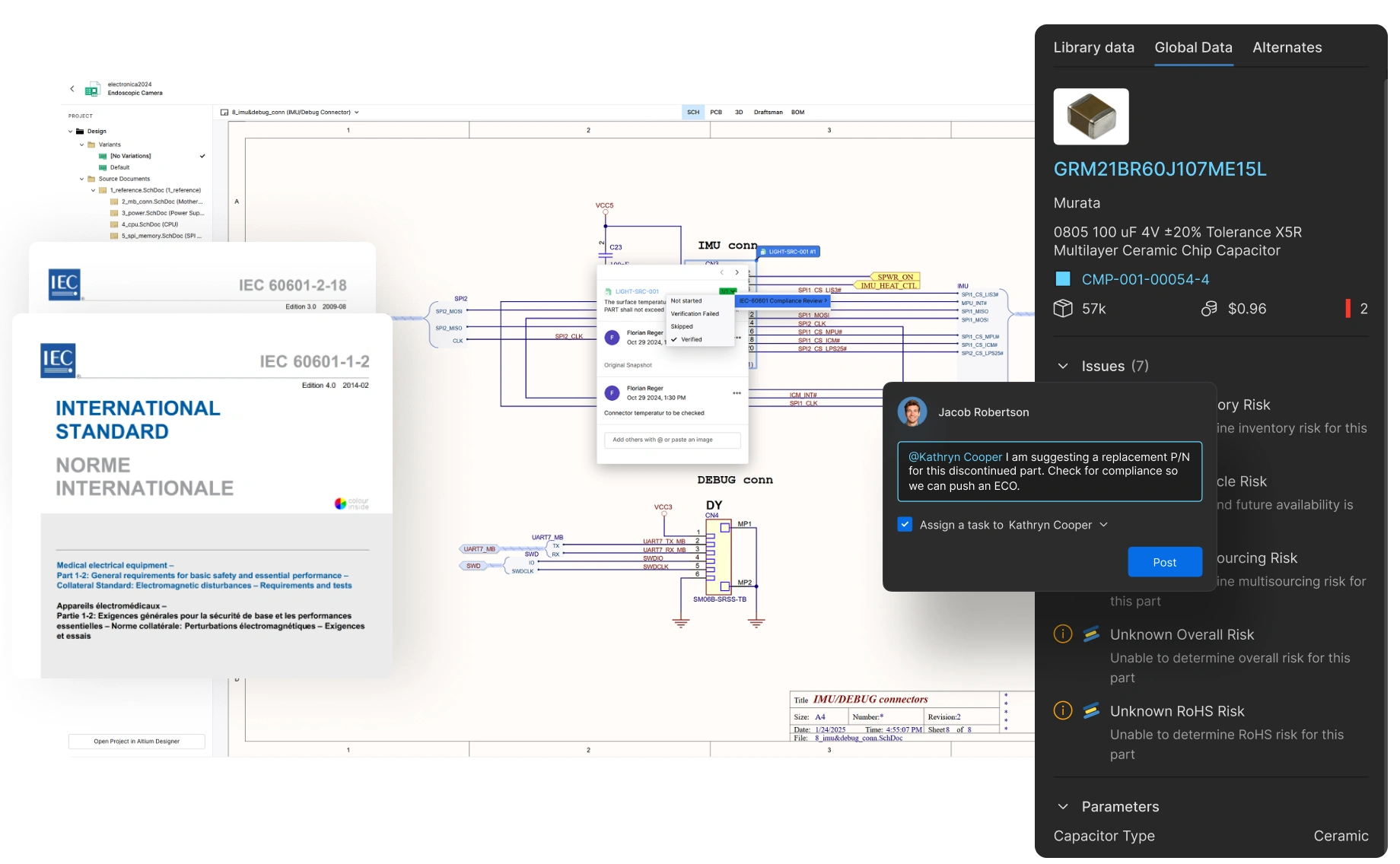
Power Collaboration
for Innovation
With real-time design sharing and coordinated workflows, accelerate problem-solving and faster response to market needs. With seamless collaboration, focus on breakthrough medical device technology, and not administrative tasks.
Accelerate Time-to-Market
Eliminate data silos and enable collaboration across engineering, quality and regulatory teams. Iterate faster and automate documentation to reduce approval timelines, delivering competitive market advantage.
Secure Long-Term
Component Availability
Safeguard the longevity of medical devices by predicting component lifecycles during the design phase. Utilize supply analytics to align engineering with procurement, ensuring all specified parts meet regulatory standards and have guaranteed long-term availability.
Integrate Design-to-Manufacturing
Direct PLM integration eliminates manual data transfer between engineering and manufacturing. Automated BOM synchronization accelerates manufacturing readiness, reducing time to market while ensuring design integrity.
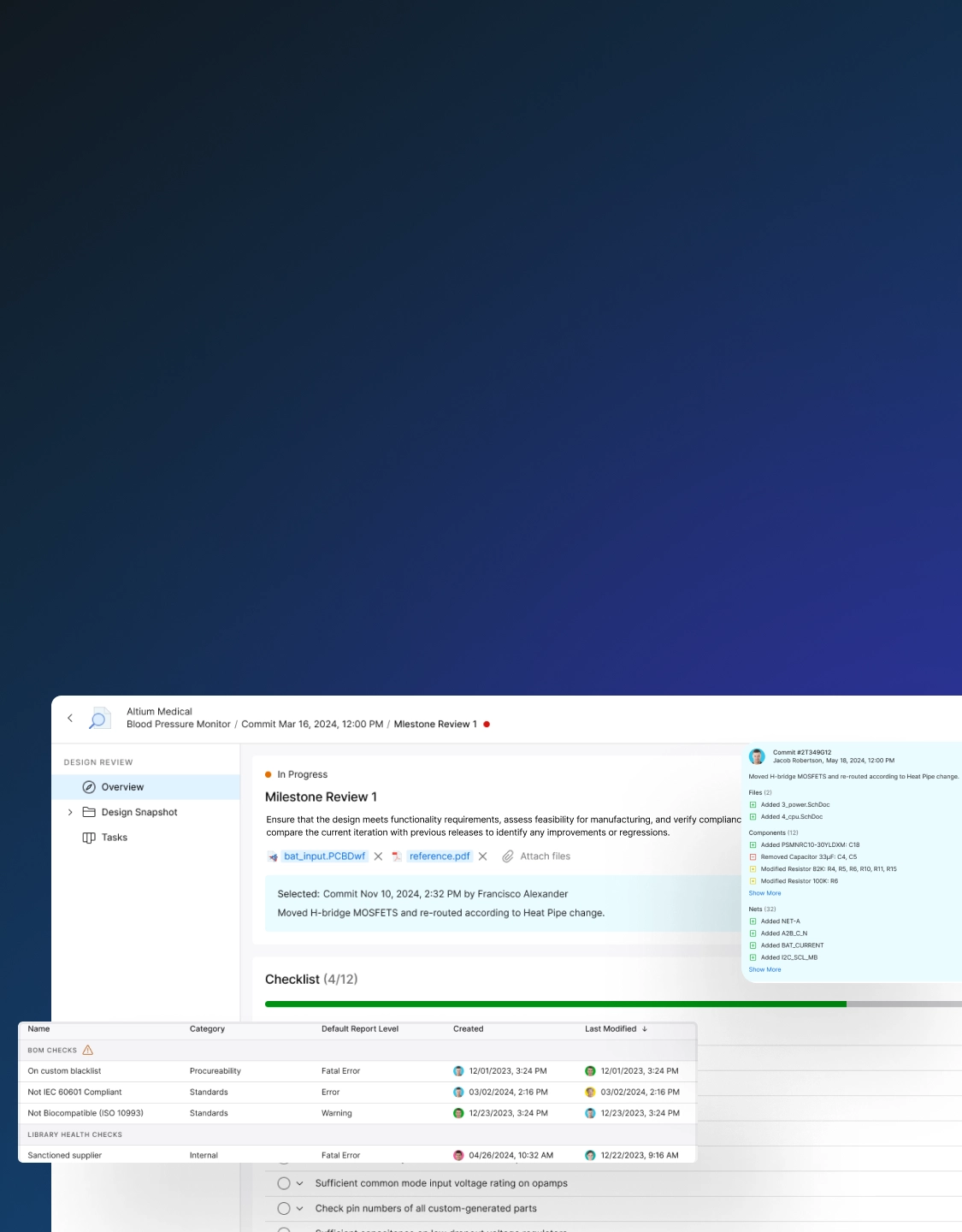
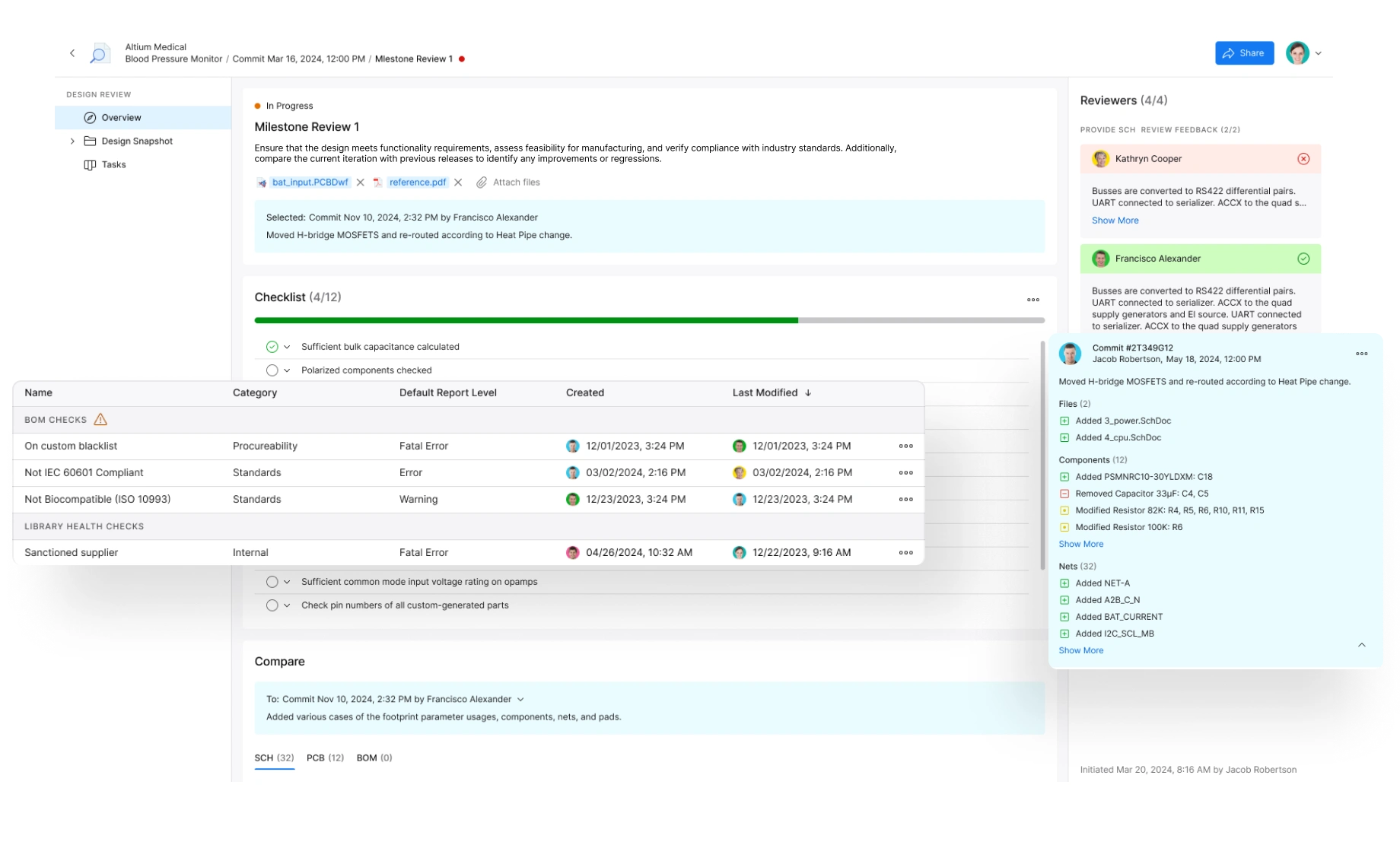
Accelerate Approvals and Ensure Compliance
Minimize regulatory risks and accelerate approvals. Automate documentation, maintain audit trails, ensure design control adherence, and provide complete traceability.
Mitigate Risk through Traceability
Complete design history with automated version control supports audit trails and regulatory compliance. Connected workflows link design decisions to risk management, reducing costly recalls and protecting brand reputation.
Scale Compliance Management
Enterprise-wide standardization ensures consistent quality and compliance across multiple projects and global teams. Controlled workflows adapt to evolving regulatory requirements, supporting business growth.
Ensure Audit-Ready Compliance
Integrated workflows and audit-ready documentation ensures verification and validation against requirements, eliminating manual documentation efforts, and allowing for the acceleration of regulatory submissions.
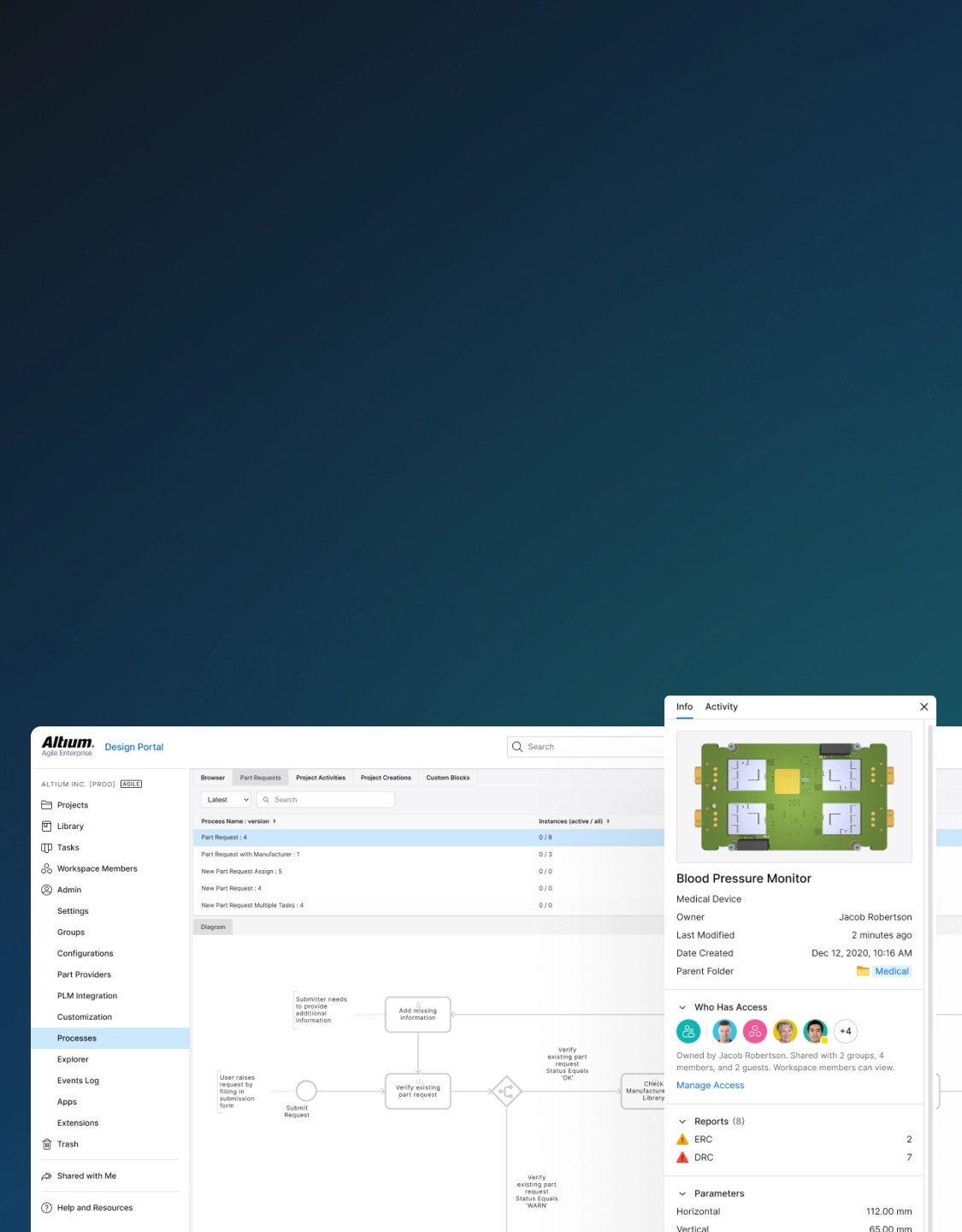
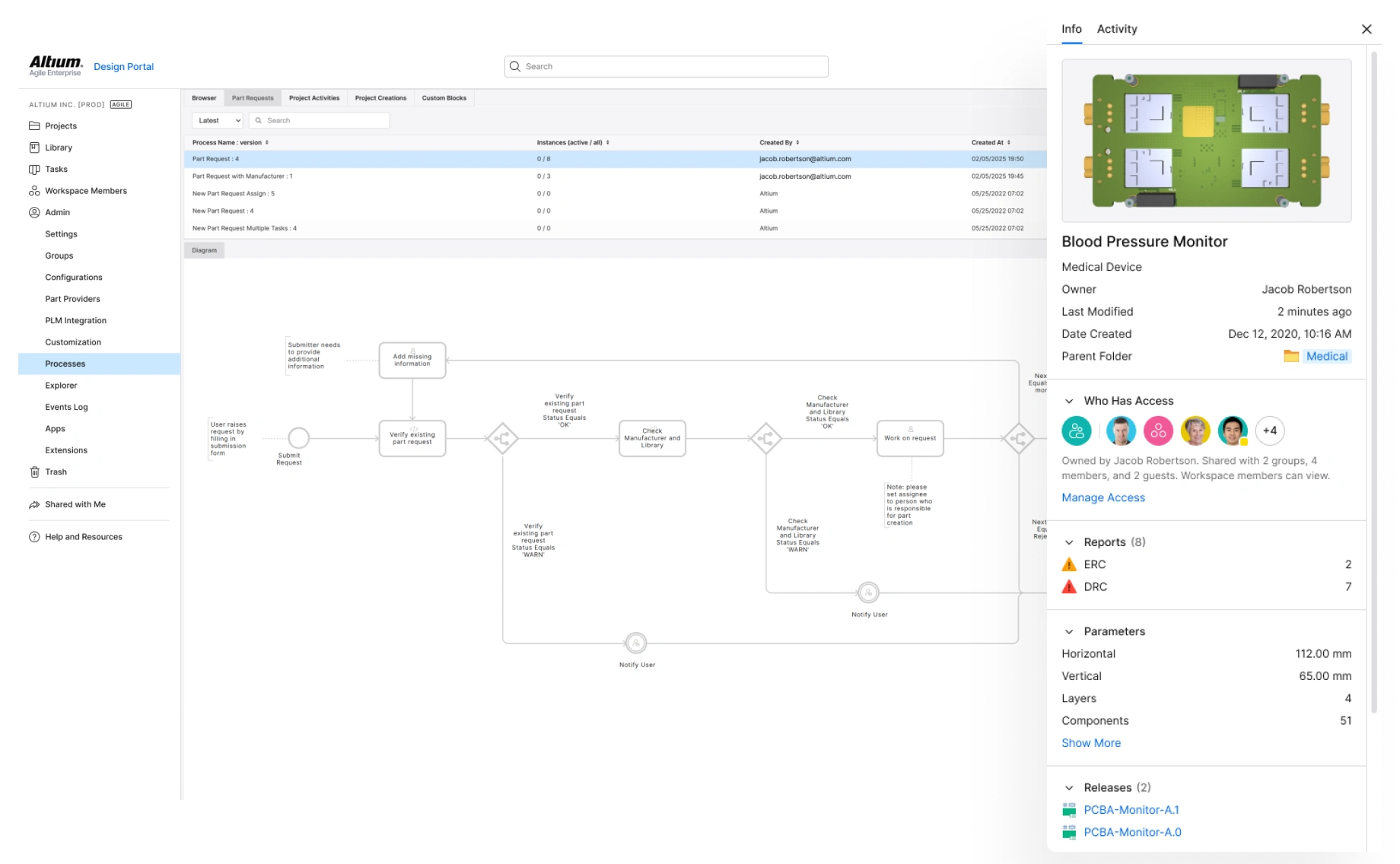
Manage Complex Electronics
System-level control of complex medical device electronics ensures safety, interoperability, and regulatory compliance. Manage complex designs, and standardize component selections across subsystems, and maintain the digital thread.
Get Interoperability Across Systems
A unified design environment ensures system-level performance and coordinates with embedded software, mechanical enclosures and connectivity requirements.
Reduce Complexity and Risk
Enterprise security and role-based access controls reduce complexity and risk. Supporting secure and traceable documentation as well, users can be confident that they are meeting security objectives.
Provide Security and Quality Control
System-level control of data to ensure quality, accuracy, and security. Controlled permissions ensure only authorized personnel modify critical designs and that all changes are tracked.

The Best Teams Around the
World Use Altium
Explore Other Industries

Learn More About
Altium Agile Enterprise
Platform-based solution for electronics product
and systems, delivering control without complexity
Complete this form and we will reach out to answer your questions about Altium Agile Enterprise.









